Pterocarpus marsupium and Gymnema Sylvestre Powder in Alleviation of Biochemical Abnormalities Associated with Human Type 2 Diabetes Mellitus- Juniper Publishers
Juniper Publishers- Journal of complementary medicine
Abstract
Objectives: This observational study investigated the antihyperglycemic, antihyperlipidemic and antioxidant potential of Pterocarpus marsupium (Vijaysaar) and Gymnema sylvestre (Gudmar) (“VG”) Therapy in patients with type 2 diabetes mellitus.
Methods: Subjects with type 2 diabetes
mellitus were administered a phytochemical formulation consisting 50 ml
of aqueous extract derived from 3 gm “VG” powder was administered twice a
day for six months. The blood glucose levels were monitored at monthly
intervals; glycosylated hemoglobin, lipid profile and biomarkers of
oxidative stress, liver and kidney functions were monitored at three
monthly intervals.
Results: Daily administration of the “VG”
Therapy regularly for 6 months resulted in significant reduction of
blood glucose and glycosylated hemoglobin levels. There was also a
significant increase in high-density lipoprotein cholesterol levels with
concomitant decrease in total cholesterol, triglyceride, low-density
lipoprotein cholesterol, and very-low-density lipoprotein levels.
Significant improvement in the biochemical markers of oxidative stress
was recorded.
Conclusion: TThe results suggest that herbal
formulation “VG” is beneficial in improving glucose and lipid
homeostasis in type 2 diabetics and may serve as an adjunct therapy.
Keywords: Hyperglycemia; Dyslipidemia; Oxidative stress; Type 2 diabetes mellitus; Pterocarpus marsupiu; Gymnema SylvestreAbbrevations: BMI: Body Mass Index; DBP: Diastolic Blood Pressure; FPG: Fasting Plasma Glucose; GSH: Reduced Glutathione; HbA1c: Glycosylated Haemoglobin; HDL: High-Density Lipoprotein Cholesterol; LDL: Low-Density Lipoprotein Cholesterol; PPG: Postprandial Glucose; SBP: Systolic Blood Pressure; SOD: Superoxide Dismutase; SGOT: Serum Glutamate Oxaloacetate Transaminase; SGPT: Serum Glutamate Pyruvate Transaminase; TBARS: Thiobarbituric Acid Reacting Substances; VLDL: Very low-density lipoprotein cholesterol; VG: Vijaysaar and Gurmar
Introduction
Type 2 diabetes mellitus is reaching epidemic
proportions worldwide and is a complex metabolic disorder characterized
by hyperglycemia and dysregulated lipid metabolism [1,2]. Abdominal
obesity and dyslipidemia (increased triglycerides, high LDL-cholesterol
and low HDL-cholesterol), and hypertension, as well as altered platelet
function [3] are the major risk factors contributing to type 2 diabetes.
The metabolic defects that underlie the development of type 2 diabetes
may include islet β-cells dysfunction, hyperglucagonemia, elevated
endogenous glucose production, development of peripheral insulin
resistance, inflammation of adipose tissue, and dysregulation of
adipokines production [4]. Chronic elevation of blood glucose which is a
central factor in the production of
reactive oxygen species (ROS) [5] which in turn promote cellular damage
and contribute to the development and progression of diabetic
complications, such as cardiovascular disease, nephropathy, retinal
blindness, neuropathy, and peripheral gangrene [6]. The conventional
allopathic drugs, sulfonylureas, meglitinides, biguanides, metformin,
thiazolidinediones, miglitol, acarbose, etc. although effective [7], are
associated with minor or major side effects on long term use [8].
Statins used in the treatment of dyslipidemia have their own limitations
[9].
Plant based Indian traditional medicines such as
Ayurveda have been used since ancient times in the treatment of diabetes
[10]. Ethnobotanical studies of traditional herbal medicine used for
diabetes have identified more than 1,200 species of plants with
hypoglycemic activity [10]. Medicinal plants are frequently
considered to be less toxic and free from side effects than
synthetic ones. The antidiabetic effects of several plant extracts
and herbal formulations and their bioactive compounds have
been identified and characterized for the treatment of diabetes
mellitus [11,12].
Polyherbal formulations [13], have been shown to exhibit
antidiabetic, antihyperlipidemic and antioxidant potential
in animal models as well as in diabetic patients [14]. The
phytochemical based formulations consisting of multiple
herbs are liable to produce a large number of metabolites that
may act on multiple targets in the body and hence, polyherbal
formulations (Which are used in traditional practice) are
preferred over mono therapeutic ones. Although, phytochemical
based formulations have extensively been used, studies on their
proof of efficacy are limited [15].
Gymnema sylvestre (Gudmar) is a climber grown in tropical
forests of South-East and belongs to the family Asclepiadaceae
G. sylvestre is considered to have a great anti-diabetic potential
and used in several anti-diabetic formulations. There are several
reports in animal models as well as in diabetic patients which
shows that extract of G. sylvestre leaves have anti-diabetic
potential [16,17], and these effects are attributes due to increase
in insulin secretion [18]. Pterocarpus marsupium (Vijaysaar)
is the most versatile plant with a wide spectrum of biological
activities and belongs to the family Fabaceae. The P. marsupium
is reported to be anti-diabetic and associated with anti-obesity,
anti-hyperlipidemic, antiinflammatory, anti-oxidative, antitumorigenic
functions [19-21]. The aim of this study is to explore
the therapeutic potential of combined effects of the regular
consumption of Pterocarpus marsupium and Gymnema sylvestre
in human subjects with type 2 diabetes mellitus.
Materials and Methods
Study sample
A total of 45 type 2 diabetic patients attending the weekend
diabetes clinic run by the Centre for Translational Research,
School of Studies in Biochemistry, Jiwaji University, India
expressed their willingness to participate in the study. Of these,
35subjects met the inclusion criteria who were then explained
the necessity of maintaining defined lifestyle pattern during the
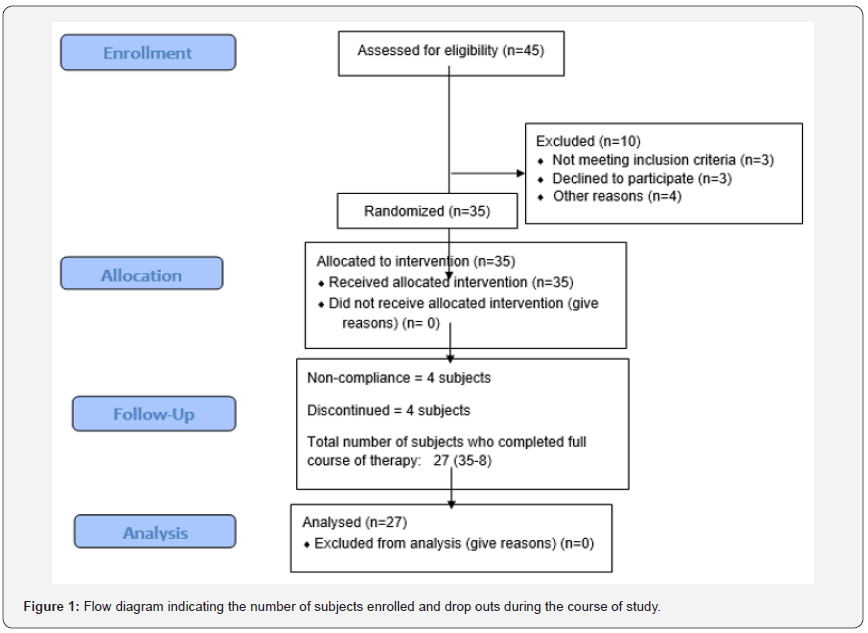
course of the study. Out of 35 subjects, 8 subjects were eliminated
during the course of the study due to non-compliance. The
remaining 27 subjects took the drug regimen regularly as per
the experimental design. Figure 1 shows the number of subjects
enrolled and drop outs during the course of study. The patients’
baseline characteristics are summarized in (Table 1).
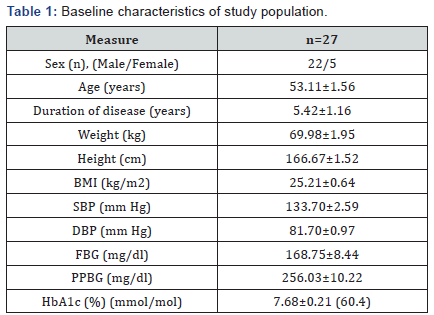
Data are expressed as Mean ± SEM;
BMI-Body mass index; SBP- Systolic blood pressure; DBP- Diastolic
blood pressure; FBG-Fasting blood glucose; PPBG-Post prandial
blood glucose; HbA1c: Glycosylated Haemoglobin.Study design
The study design included following steps:
a) Selection of patients meeting inclusion criteria and
those consented to participate in the study.
b) Measurement of anthropometric and biochemical
parameters once before and at selected intervals during the
course of the study.
c) Administration of “VG” Therapy in defined dose daily
for a period of 6 months.
d) Measurement of parameters at the end of the study.
e) Analysis of the data.
Inclusion criteria
a) Non-insulin dependent diabetes diagnosed, as per the
criteria of the World Health Organization;
b) Both genders between the ages of 30-65 years;
c) Body Mass Index in the range of 18.5 to 40;
d) Participants who understood the benefits of the study
and signed a written informed consent;
Exclusion criteria
a) Presently using other blood glucose regulating agents;
b) Daily intake of alcoholic beverages;
c) Smokers consuming more than 1 pack/day;
d) Patients diagnosed as type I and insulin dependent
type II diabetics;
e) Patients with hepatic or renal disease, pancreatitis,
cardiac problems, uncontrolled hypertension, malnutrition
and severe immune deficiency.
f) Non-compliance during the course of study.
Before starting the study all participating subjects were
given a verbal explanation about the objectives of the study,
nature of drug formulation (Kwath) to be consumed daily, (“VG”
Therapy) rationale and duration of therapy in local language.
They were asked to avoid a carbohydrate rich diet and advised
regular walking for about 4-5 km during the course of study.
The patients who consented to participate in the study were
registered, anthropometric measurements; weight, height and
waist were recorded at the beginning and at selected intervals
during the study period. The patients were kept exclusively on
“VG” therapy.
Composition of drug (VG) formulation
The “VG” formulation consisted of a mixture of Pterocarpus
marsupium (Vijaysaar), and Gymnema sylvestre (Gurmar) (Table
2) and was provided by the M/S Dindayal Industries Ltd. India.
The individual plants were identified by Prof. A.K. Jain, School
of Studies Botany, Institute of Ethnobiology Jiwaji University
Gwalior. Voucher specimens were prepared and deposited at the
Centre (IOE-501 and IOE-505).

Composition of VG formulation: 1:1.
Preparation of VG kwath
3 gm of VG powder was suspended in 100 ml drinking water,
kept overnight, boiled till 50% water is evaporated, cooled to
room temperature and consumed on an empty stomach twice
a day for six months under the supervision of an Ayurvedic
Physician.
Biochemical parameters
The fasting and postprandial plasma glucose levels
were monitored at monthly intervals, while the glycosylated
hemoglobin (HbA1c), biomarkers of oxidative stress viz.,
Superoxide dismutase (SOD), catalase, reduced glutathione
(GSH), Thiobarbituric Acid Reactive Substances (TBARS) and
lipid profile, biochemical markers of kidney and liver functions
were monitored at baseline, at the middle (3 month) and at the
end (6 month) of the therapy.
Fasting and postprandial plasma glucose was estimated
by the Glucose oxidase/Peroxidase method [22]. Glycosylated
hemoglobin (HbA1c) was estimated by the ion exchange
resin method [23]. Estimation of plasma total cholesterol by
the Cholesterol oxidase Phenolaminophenazone CHOD-PAP method [24], triglyceride by GPO-PAP method [25], High density
lipoprotein cholesterol (HDL) by Polyethylene glycol/CHOD-PAP
method [26], Low-density lipoprotein cholesterol (LDL) and
Very-low density lipoprotein cholesterol (VLDL) were calculated
by the Friedewald formula, urea by the modified Berthelot
method [27], uric acid by uricase/PAP method [28], creatinine
by modified Jaffe’s kinetic method [29], Alanine transaminase
(SGPT or ALT) and serum Aspertate transaminase (SGOT or AST)
by the modified International Federation of Clinical Chemistry
method [30], and bilirubin by method of L. Jendrassik [31] was
assayed using standard kits from Crest Biosystems, Goa (India).
Markers of oxidative stress such as super oxide dismutase (SOD)
Winterbourn et al. [32], catalase by Sinha, [33], glutathione (GSH)
Ellman et al. [34], and Thiobarbituric Acid Reactive Substances
(TBARS) Ohkawa et al. [35] were checked.
Ethical clearance
The study protocol was duly approved by the Institutional
Human Ethics Committee (JU/IHEC/2013-A/13).
Statistical analysis
Statistical analysis was carried out using a paired t test
(Sigma stat 3.5). A p-value *p<0.05, **p<0.001 was considered
statistically significant.
Results
Effect of VG therapy on hyper-glycemia
Table 3 shows the fasting and postprandial blood glucose
levels at monthly intervals in subjects on “VG” therapy. A
significant decrease was recorded for fasting (13.5% P<0.05),
postprandial (15.0%, <0.001) glucose levels and glycosylated
hemoglobin (9.8%) at the end of six months therapy.

Data are expressed as Mean ± SEM; *p<0.05; **p<0.001 compared to Base line values
HDL-High-density lipoprotein cholesterol;
LDL-Low-density lipoprotein cholesterol; VLDL- Very- low density
lipoprotein cholesterol; TC-Total
cholesterol
Effect of VG therapy on lipidemia
Table 4 shows the results of the lipid profile of patients on
“VG” therapy. Total cholesterol, triglycerides, LDL and VLDL were
significantly decreased after six months therapy by 9.6%, 14.4%,
15.7%, and 14.4% respectively (P<0.05). HDL cholesterol was
elevated from32.50±1.11 to 35.69 ± 1.02 (P<0.001).
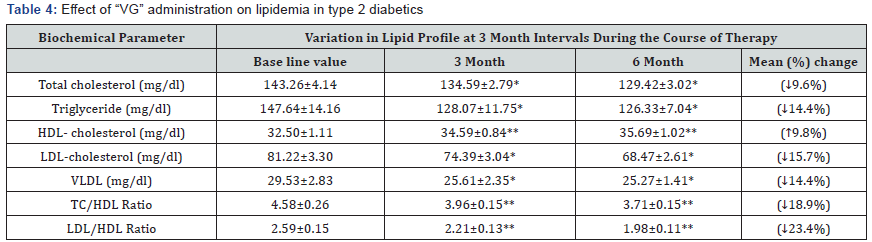
Data are expressed as Mean ± SEM; *p<0.05; **p<0.001 compared to Base line values
HDL-High-density lipoprotein cholesterol;
LDL-Low-density lipoprotein cholesterol; VLDL- Very- low density
lipoprotein cholesterol; TC-Total
cholesterol
Effect of VG therapy on biomarkers of oxidative stress
Significant (P<0.05, P<0.001), improvements in GSH level
(from 1.98±0.17 to 2.59±0.21 mg/dl), SOD activity (from
0.63±0.05 to 0.94±0.08 μM/min/mg protein), catalase activity
(from 7.64±0.24 to 9.44±0.23 μM/min/mg protein) were
recorded. A significant decrease (P<0.001) in TBARS (from 457.19±8.09 to 415.15±7.47 (moles of Malondialdehyde/ml of
blood) were recorded at the end of six months therapy.
Effect of VG therapy on markers of toxicity
The effect of “VG” therapy on kidney function was monitored
by estimating urea, uric acid and creatinine levels in plasma
at specified intervals during the course of therapy. The data
presented in Table 5 showed significant (P<0.05), reductions
in Urea (from 29.97±1.20 to 27.10±0.88mg/dl) and uric acid
(from 5.08±0.20 to 4.67±0.20 mg/dl) and notable reduction
in creatinine (from 0.78±0.06 to 0.73±0.05) at the end of six
months therapy.
Data are expressed as Mean ± SEM; *p<0.05; **p<0.001 compared to Base line valuesGSH: Reduced glutathione; SOD-Superoxide dismutase; TBARS- Thiobarbituric acid reacting substances.
SGOT- Serum Glutamate Oxaloacetate Transaminase; SGPT- Serum Glutamate Pyruvate Transaminase.
Significant (P< 0.05), variations in biochemical markers of
liver functions, namely bilirubin (from 0.83±0.06 to 0.70±0.04
mg/ml) and SGOT (from 22.47±1.61 to 18.00±1.04 IU/L) as well
as SGPT (from 23.60±2.32 to 19.26±1.38 IU/L), at the end of six
months was observed (Table 6).
Effect of VG therapy on hypertension and body mass index
Table 7 shows variations in systolic blood pressure (from
133.70±2.59 to 129.22±2.39 mmHg), diastolic blood pressure
(from 81.70±0.97 to 79.56±0.93 mmHg) and body mass index
(from 25.21±0.64 to 24.22±0.60 kg/m2) at the end of the six
months therapy.

Discussion
Currently, plant based therapies (main or supplementary)
are being used globally for the management of type 2 diabetes.
In India, the indigenous systems of medicine viz., Ayurveda,
Siddha & Unani employ plant based extracts for treatment of
diabetes (Premeha in Ayurveda). Most of the Ayurveda based
formulations depend on plants/herbs based decoctions, and
powders. In the present study, administration of the aqueous
extract of VG that consisted of equal amounts of the ingredients
for over a period of 6 months to type 2 diabetic subjects resulted
in significant improvements of altered biochemical markers. Significant alleviation in glucose homeostasis, as marked by
noticeable changes in glycosylated hemoglobin, blood glucose
levels, and lipid profile were observed. The G. sylvestre leaf
extracts are reported to reduce hyperglycaemia in experimental
animals [16], and human type II diabetics [17]. The G. sylvestre is
reported to promote insulin secretion, probably by regeneration
of pancreatic beta cells [17,18]. The gymnemagenin and
gymnemic acids in G. sylvestre, showed to exhibit potent
antioxidant, and hepatoprotective activities [36]. Ethanolic
extract of G. sylvestre was reported to possess anti‑apoptotic
potential in STZ‑induced diabetic cardiomyopathy, and affected
the levels of leptin, insulin, dyslipidemia, apolipoproteins, lipids,
LDH (serum lactate dehydrogenase) in experimental models
[37]. Administration of deacylgymnemic acid DAGA (200mg/kg),
a derivative of gymnemic acid reduced Systolic blood pressure
and significantly improved the fasting plasma glucose and
HOMA-IR (homeostatis model assessment insulin resistance)
with marginal improvement in lipid profile [38]. The G. sylvestre
is also reported to inhibit absorption of glucose from intestine.
The atomic arrangement of gymnemic acid molecules is similar to
that of glucose molecules and they reduce the blood glucose level
by preventing the absorption of sugar molecule in the intestine.
It is attributed by filling the receptor location in the absorptive
external layer of the intestine by gymnemic acid molecules [39].
In a controlled study, an aqueous extract of the leaves of G.
sylvestre, (GS4) when administered at the dose of 400mg/day,
to subjects with insulin-dependent diabetes mellitus (IDDM),
the insulin requirement came down together with fasting
blood glucose, HbA1c and serum lipid levels soon after therapy.
However, the GS4 administration failed to check glycosylated
haemoglobin and glycosylated plasma protein levels on long
term follow up. The GS4 therapy is reported to enhance insulin
secretion rate, either by regeneration or revitalisation of the beta
cells [40].
The effectiveness of GS4, further investigated as a
supplementary drug to conventional oral anti-hyperglycemic
drugs. Administration of GS4 (400mg/day) to 22 Type 2 diabetic
patients receiving conventional therapy resulted in significant
reductions in blood glucose and, glycosylated hemoglobin level.
In addition, five of the 22 diabetic patients showed improved
glycemic control with GS4 alone and were discontinue their
conventional drug [17].
In another study, supplementation of the diet with G. sylvestre
at the dose of 500 mg per day for a period of 3 months reduced
polyphagia, fatigue, blood glucose levels with concurrent
reductions in HbA1c, lipid profile suggesting beneficial effect
of G. sylvestre supplementation in the management of diabetes
mellitus [41].
Pterocarpus marsupium is traditionally used in Indian
ayurvedic medicine for the treatment of diabetes and has
been shown to control diabetes in experimental animals [42].
The phenolic C-glycosides present in P. marsupium regulated
glucose homeostasis and significantly decreased the TNF-α
level in experimental diabetes [43]. The phenolics, marsupsin,
pterosupin, pterostilbene and (-)-epicatechin present in P.
marsupium have been identified as the blood sugar lowering
components [20,44].
The alcohol extract of the bark of P. marsupium Roxb
shown to exhibit potent antidiabetic activity and corrected the
metabolic alterations in diabetic rats [42]. Aqueous extracts of P.
marsupium Linn significantly attenuated hyper insulinaemia as
well as hypertriglyceridaemia and also prevented the alteration
in metabolic patterns [45]. The Indian Council of Medical
Research undertook an anti-diabetic phase II open trial at four
centers across Indian using Vijaysar (Pterocarpus marsupium).
Vijaysar was tested in newly-diagnosed or untreated non-insulin
dependent diabetes mellitus (NIDDM) patients between 35 and
60 age for 12 weeks. Among the 93 patients who completed 12
wk of treatment, both the fasting and postprandial blood glucose
levels fell significantly (P < 0.001), by 32 and 45mg/dl at 12 wk
from the initial means of 151 and 216mg/dl respectively. Mean
HbA1c Other laboratory parameters remained stable during the
designated treatment period of 12 wk. Also, no side-effects were
reported [46].
Our previous study done with the polyherbal formulation,
“GSPF kwath” which contains Vijaysar and Gurmar as two of
the ingredients has shown antidiabetic and antioxidative effects
[13]. “GSPF kwath”, showed 23.5%, 26.7% and 11.7% reductions
in fasting, postprandial blood glucose and HbA1c respectively. In
the present study “VG” containing only two of the components
of GSPF proved highly beneficial in restoring not only hyperglycemia
but also lipidemia and antioxidative potential. The
changes observed on glycemic, lipidemic and oxidative stress.
Oral administration of “VG” therapy for 6 months had no adverse
effects, either on kidney or liver functions and in fact showed a
significant improvement in the functioning of these vital organs.
Conclusion
In conclusion, the “VG” administration to type 2 diabetic
patients with varying degrees of glycemia, lipidemia and
oxidative stress provide an efficient alternative to conventional
antidiabetic drugs when coupled with active lifestyle and dietary
changes. Significant decrease in biochemical markers viz., blood
glucose, HbA1c, total cholesterol, triglycerides, LDL-cholesterol,
and VLDL-cholesterol and higher values of HDL, following VG
therapy are testimony to the anti-diabetic efficacy of VG. Further,
the formulation did not show any adverse effects on liver and
kidney functions and may be a potential natural and safe
therapy for treatment and prevention of diabetic complications
but randomized controlled trials are required to confirm these
findings.
Acknowledgment
We thank M/S Dindayal Industries Ltd., Gwalior (M.P.) for
the supply of the Vijaysaar and Gurmar powder and the financial
support from AYUSH, New Delhi, (F.NO. Z.31014/02/2009/
EMR-CCARS) is duly acknowledged. We are grateful to all the human subjects who volunteered to participate in the study.
Our profound thanks extend to colleagues of the laboratory and
students for their unreserved support in conducting this study.



Comments
Post a Comment